Neurotransmitters mediate communication between neurons at specific connection sites referred to as synapses. The catecholamines dopamine and noradrenaline are neurotransmitters that control many brain functions such as decision making, motivation, learning and body movements. Many studies have found a connection between altered levels of these transmitters and several psychiatric and neurological disorders like Parkinson’s disease, ADHD and schizophrenia.
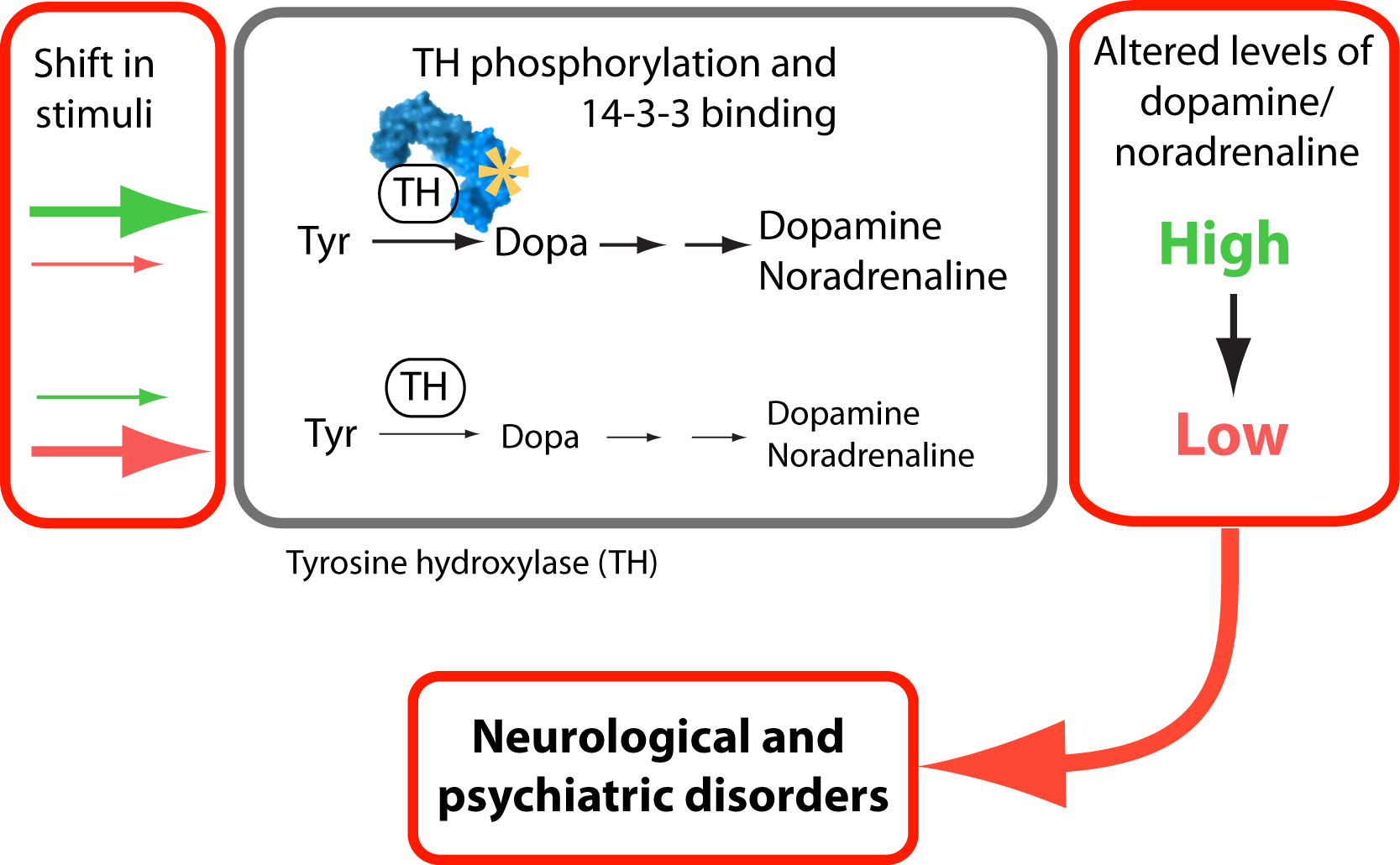
The first enzyme in the biosynthesis of catecholamines is called tyrosine hydroxylase (TH). This enzyme regulates the level of catecholamine production in neurons. The activity of TH is controlled at many different ways, one of which is by phosphorylation. Phosphorylation of enzymes is a common mechanism to regulate their activity in response to signals sensed by the cells. This process is called signal transduction, and different signal transduction pathways can regulate the activity of different enzymes within the cell by reversibly adding a phosphate group to one or more amino acid residue of the enzyme.
In a study that involved researchers in the Neurotargeting- and Biorecognition groups, we present new insight into the regulation of TH by phosphorylation. We describe details of how phosphorylation of TH at a specific site leads to the formation of regulatory protein complexes. We characterized the role of the 14-3-3 protein family in regulating TH. There are seven different 14-3-3 proteins found in humans, but we do not know which one of these are the most active in regulating TH. In our study we compared different 14-3-3 protein types in their binding to and regulation of TH. Our results showed that TH formed complexes with all the 14-3-3 proteins, but there were differences in the potency of enzyme activation. We also compared the 14-3-3 proteins for their ability to control TH phosphorylation homeostasis.
This knowledge will give us more detailed overview over the regulation of the catecholamine biosynthesis that can be used further in understanding of defected neuronal pathways and designing the drugs to overcome these defects.
By Sadaf Ghorbani (PhD student)