Nevrotransmittere er signalmolekyler som nerveceller bruker for å kommunisere. Katekolaminer er en familie av nevrotransmittere som inkluderer dopamin og noradrenalin. De fungerer med å justere hvilke kommunikasjonsveier i hjernen som påvirker hverandre mest. På den måten er de med på å kontrollere mange forskjellige hjerneaktiviteter, som bevegelser, konsentrasjon, hukommelse, læring, stress og avhengighet.
Endringer i disse nevrotransmitterene er observert i mange forskjellige nevrologiske og psykiatriske lidelser og mye av medisineringen innenfor psykiatrien virker ved å endre balansen i disse signalstoffene. ADHD medisiner virker f.eks ved å forlenge signalet fra dopamin og noradrenalin. I den nevrodegenerative sykdommen Parkinsons sykdom, er det en spesifikk gruppe nerveceller som lager og skiller ut dopamin som dør. Dette påvirker i særlig grad systemene som er involvert i styring av bevegelse.
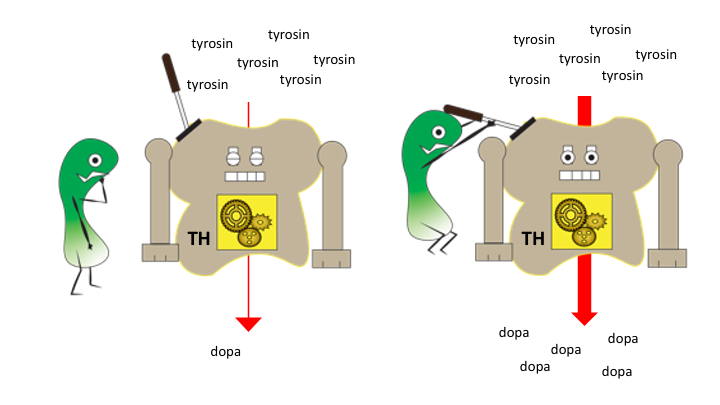
Dopamin og noradrenalin lages fra aminosyren tyrosin, gjennom flere kjemiske reaksjoner som alle utføres av enzymer – biologiske katalysatorer. Det første steget omdanner tyrosin til dopa ved help av enzymet tyrosinhydroksylase (forkortet TH). Produktet som TH lager, dopa, er også kjent som en forbindelse som blir brukt til medisin i Parkinsons sykdom (kjent som L-Dopa) og for pasienter som har defekt TH aktivitet – TH Deficiency.
Reaksjonen som TH katalyserer er et viktig kontrollpunkt for nivået dopamin og noradrenalin. Slike kontrollpunkter i metabolismeveier styres av cellens signalveier. På den måten kan cellene regulere syntesen, slik at den er tilpasset aktiviteten til nervecellen. I en ny publikasjon fra nevrotargeting gruppen og Helse Bergen, beskrives nye måter aktiviteten til TH enzymet kan styres gjennom signalprosesser. Disse signalprosessene skrur av og på ulike ”brytere” på enzymet og dette endrer enzymet sine egenskaper og effektivitet. Studien viser at en av bryterene på TH kan styre tilgangen til andre brytere som direkte styrer hvor effektivt enzymet arbeider. Denne forståelsen kan hjelpe oss til å forstå hvordan katekolaminene er regulert og for å utvikle nye medisiner for sykdommer hvor disse signalsystemene er involvert.
Av Sadaf Ghorbani og Rune Kleppe